Coronavirus vaccine: Final stage of Oxford trial paused after participant falls ill
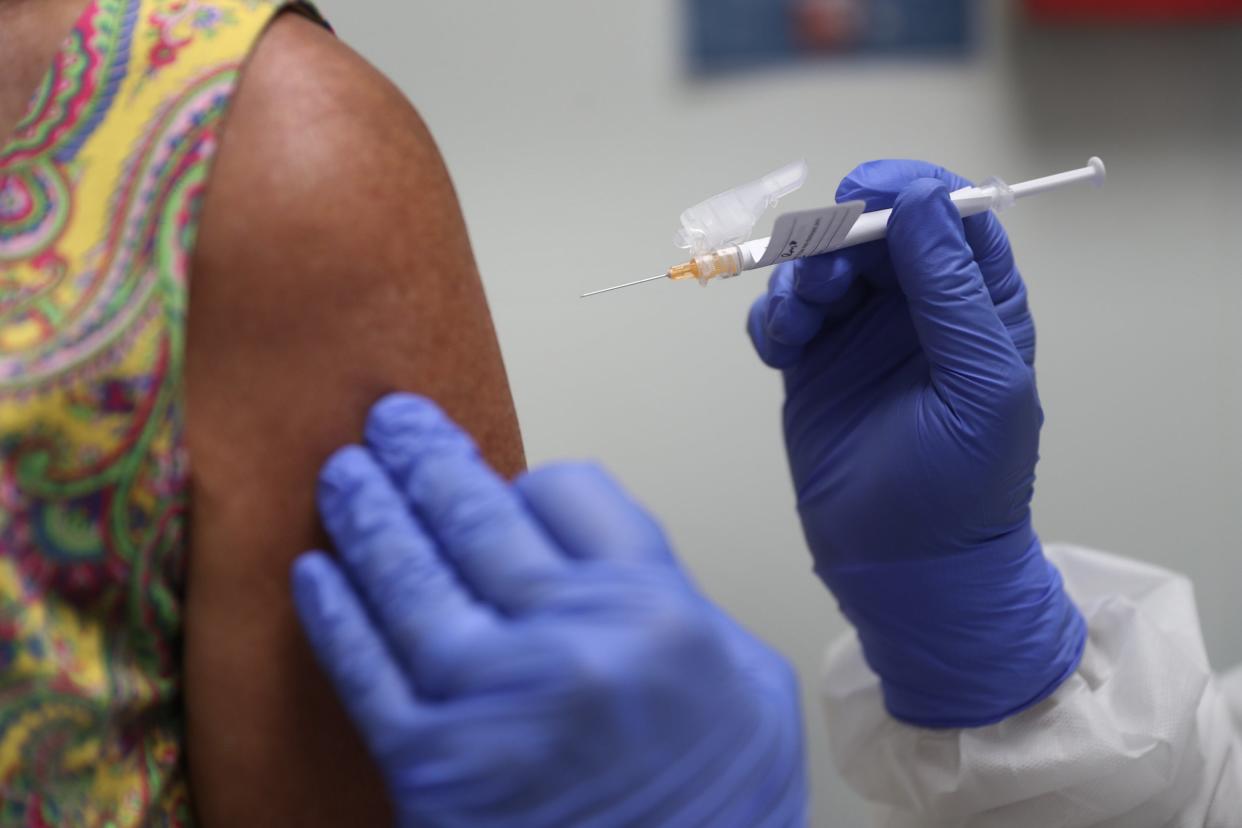
The clinical trial of a high-profile coronavirus vaccine from AstraZeneca has been paused after one of the UK participants suffered a “potentially unexplained illness”, according to a company statement.
While the participant had a suspected “serious” adverse reaction, it was possible the illness was not caused by the vaccine, according to the statement first reported by Stat.
“As part of the ongoing randomised, controlled global trials of the Oxford coronavirus vaccine, our standard review process triggered a pause to vaccination to allow review of safety data,” the statement said.
“This is a routine action which has to happen whenever there is a potentially unexplained illness in one of the trials, while it is investigated, ensuring we maintain the integrity of the trials.”
Quoting two individuals familiar with the matter, researchers were told to place the trial on hold out of “an abundance of caution” while the finding was having an impact on other trials underway by AstraZeneca and other vaccine manufacturers, according to Stat.
The UK is among those that have signed up to buy millions of doses from AstraZeneca if they develop a successful vaccine, which is currently in Phase 3 trials at dozens of sites across the UK, the United States, Brazil and South Africa.
Developed in partnership with the University of Oxford, the AstraZeneca vaccine is backed by the US government’s Operation Warp Speed to accelerate development with $1.2bn (£905m) in aid to secure 300 million doses of a successful candidate.
Nine vaccine makers issued a statement on Tuesday pledging they would only submit vaccines for approval after demonstrating safety and efficacy, with AstraZeneca being the first major report of a serious issue from the few coronavirus trials in Phase 3.
The AstraZeneca spokesperson said the illness was a “single event” and they were committed to the safety of the participants and their trials.
“In large trials illnesses will happen by chance but must be independently reviewed to check this carefully,” the spokesperson said.
“We are working to expedite the review of the single event to minimize any potential impact on the trial timeline.”
Read more
Coronavirus: Russia’s vaccine induces immune response and is safe, early trial results suggest
Coronavirus vaccine: GSK and Sanofi start human trials
Kamala Harris says she would not trust Trump’s word alone on a coronavirus vaccine

 Yahoo News
Yahoo News 
