Novavax Covid vaccine submitted to UK regulator for approval
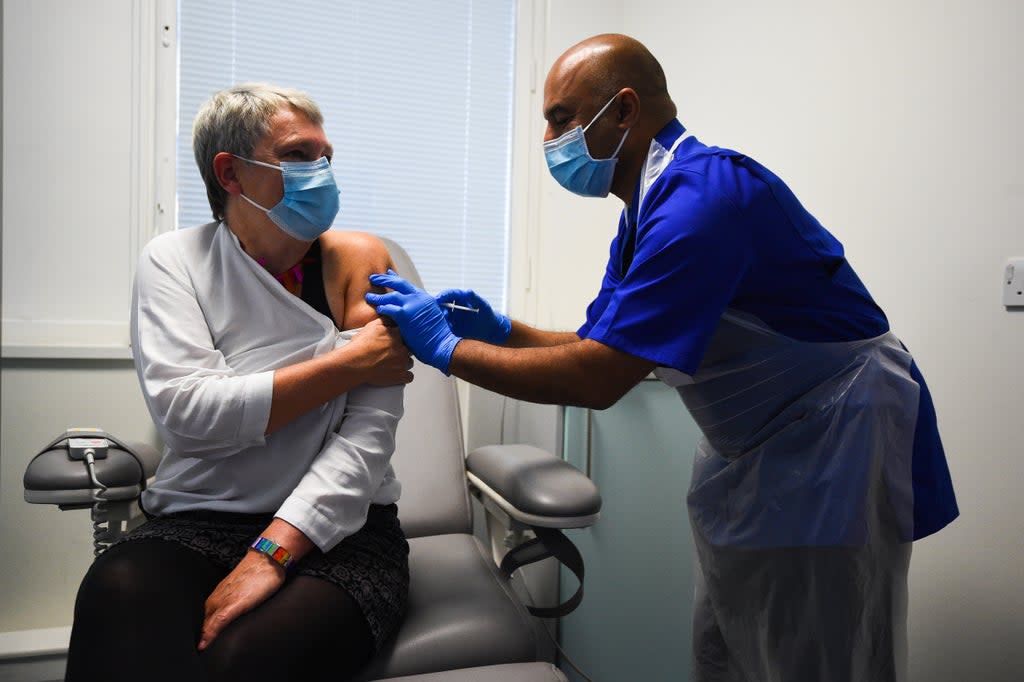
The final data on the Novavax Covid vaccine has been submitted to the UK regulator for approval.
The firm behind the vaccine said it anticipates a "positive decision" following the completion of its rolling regulatory submission to the Medicines and Healthcare products Regulatory Agency (MHRA).
More than 15,000 people took part in the phase 3 Novavax trial at various hospital sites across the UK.
Novavax said its application for conditional marketing authorisation (CMA) marks the first submission for authorisation of a protein-based coronavirus vaccine in the UK.
Britain has ordered 60 million doses of the vaccine.
Stanley Erck, the company's president and chief executive, said: "This submission brings Novavax significantly closer to delivering millions of doses of the first protein-based Covid-19 vaccine, built on a proven, well-understood vaccine platform that demonstrated high efficacy against multiple strains of the coronavirus.
"We look forward to MHRA's review and will be prepared to deliver vaccine doses following what we anticipate will be a positive decision.
"We thank the clinical trial participants and trial sites in the United Kingdom, as well as the UK Vaccines Taskforce, for their support and vital contributions to this programme."
Earlier this month, it was announced that thousands of people who took part in the trial will be able to have two further doses of another vaccine to enable them to travel abroad.
The fact the jab has not yet been approved means thousands of people have been unable to travel because other countries do not recognise Novavax as a valid Covid vaccine.
After taking advice from the Joint Committee on Vaccination and Immunisation (JCVI), the Department of Health said those in England who took part in the UK-based Novavax trial should be offered two doses of the Pfizer/BioNTech jab, with eight weeks between the doses.
Scotland, Wales and Northern Ireland's health teams were expected to follow suit.
Read More
Holidaymakers will need three jabs if they want to travel next year
Get the Covid vaccine now to give London a boost
Waiting time for Covid booster jabs could be cut to 5 months

 Yahoo News
Yahoo News 
