Why homosexual behaviour in insects may be a case of mistaken identity

Sex is costly for insects. It uses up resources such as water, energy and time. Some species, like bush crickets, can ejaculate as much as a quarter of their body weight. In others, like one species of fruit fly, their sperm is actually longer than their body.
Just as for humans, insect sex also carries risks such as the transfer of infections (nearly 100 insect STIs are known), and injury from mating trauma (like from spiny or needle-like- penises), as well as increased vulnerability to attack from predators.
Yet despite this, homosexuality in insects – which carries all of the risks of sex without the evolutionary benefit of passing on genes – has been seen in more than 100 species. In some studies, over half of matings were male on male.

No real consensus has been reached on why insect homosexuality is so common, with different studies supporting or refuting particular ideas even within the same species. But my colleagues and I have produced new evidence that supports the idea that male insects that mate with other males are simply making a mistake.
There are dozens of ideas used to explain homosexual behaviour in insects, which broadly fall into two categories. Either they suggest insect homosexuality has evolved because it actually helps males have more offspring, or that it is not an evolutionary adaptation and instead occurs when males simply don’t recognise potential mates.
In the former category, scientists have suggested male homosexual activities may reduce mating competition by distracting or injuring other males, or establishing social alliances with them or dominance over them. It could also improve heterosexual performance by keeping the ejaculate primed with fresh, younger sperm, or help males practise courtship to improve sexual performance. It could even potentially fertilise females indirectly by loading sperm onto male rivals who unwittingly transfer it in subsequent matings.
On the other hand, homosexual behaviour in insects may provide no evolutionary advantage and instead be due to confusion caused by environmental factors (as when fruit flies consume alcohol in rotting fruit). Or it may be because they fail to recognise potential mates, possibly because their social conditioning has been different, as has been shown to be important in other, more complex animals. It could also be a by-product of genes that have other beneficial effects elsewhere (a phenomenon known as pleiotropy).
Mistaken behaviour
To try to gather more evidence on this question, my colleagues and I have been studying the red flour beetle (Tribolium castaneum). Because they can complete a lifecycle within a month and are pretty hardy, these insects are ideal for studying how evolution progresses through several generations. Beetles account for 25% of all insect species, and they are also a good model from which to draw more general conclusions about insects.
Homosexuality is widely reported in many beetle species. But we had noticed that male flour beetles aren’t very fussy at all, mounting females, males, dead beetles and even oats. This suggests they may simply not be very good at recognising potential mates.
To test our theory and see whether or not this behaviour really does give the beetles any kind of evolutionary advantage, we compared two groups of flour beetles, one with more males and the other with more females. The males in the male-biased group had more competition so any that successfully fertilised a female should be better at mating. This means any traits consistently displayed across the generations by males in this group should represent an evolutionary advantage.
In our experiment, we placed males from each group in an arena and gave them the choice of mating with a male or a female. We then watched nearly 300 males mating. Because of the environment we had to create for the beetles to live in, this equated to over 50 hours of watching beetles trying to have sex at 30℃ and 60% humidity.
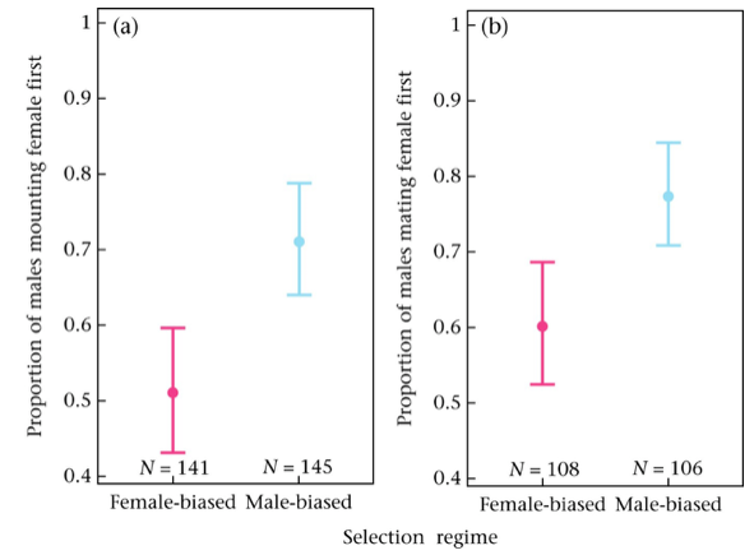
We found that the males from the two groups were equally motivated to mate. The beetles from the female-biased group weren’t picky, spending equal time trying to mate with the male as with the female. But those from the male-biased group were more likely to mate the female first, mate her more frequently and spend a greater amount of time mating her. So those beetles who came from a group with greater sexual competition were less likely to show homosexual behaviour.
This suggests the competitive males were more efficient at recognising females. This makes sense because males who aren’t as good at recognising potential mates in a group with more competition are less likely to pass on their genes. So, over multiple generations, a strong ability to recognise mates becomes more common. In the less competitive female-biased group, meanwhile, there’s less of a cost to mistakenly having sex with a male because there are more chances to mate with females and no pressure to improve the mate-spotting ability.
The implication is that homosexual behaviour in insects, especially in these beetles, isn’t an evolutionary adaptation because, where there is the pressure of sexual competition, it becomes less common over time. If it gave insects an evolutionary advantage then we’d expect it to become more common in this situation.
Instead, our research adds to the weight of evidence suggesting insect homosexuality is the result of poor mate-spotting abilities, although we still don’t know exactly why this occurs. To my knowledge, this is also the first time long-term experimental evolution has been used to study the paradox of same-sex behaviour in insects.
The results also suggest that the ability to recognise mates may require a costly amount of energy, otherwise the males in the female-biased group wouldn’t have shed this ability once they were faced with less competition and so had less need of it.
It’s also worth noting that our results were relevant to flour beetles specifically and probably to invertebrates generally. But our conclusions cannot be generalised to explain the behaviour of animals with more complex cognitive function and social structures like birds and mammals, which probably have very different reasons for same-sex mating.
This article was originally published on The Conversation. Read the original article.

Kris Sales is currently on an EnvEast DTP PhD studentship. His research was partially funded by ASAB summer studentships.

 Yahoo News
Yahoo News 