Elon Musk to start human trials with brain chip startup Neuralink

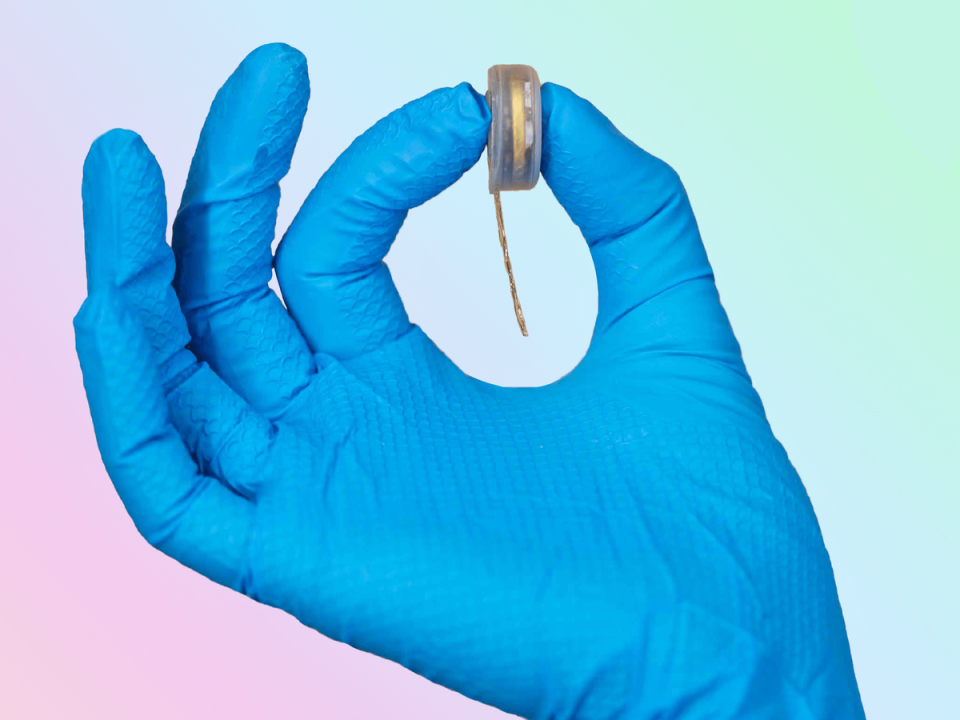
Elon Musk’s brain chip startup Neuralink has begun recruiting for its first human trials, having previously tested the technology on pigs and monkeys.
Neuralink’s brain-computer interface received approval in May from the US Food and Drug Administration (FDA) to test its technology on humans, which the company hopes can help people with brain disorders and spinal injuries.
“We’re excited to announce that recruitment is open for our first-in-human clinical trial,” Neuralink posted on X, formerly known as Twitter.
“If you have quadriplegia due to cervical spinal cord injury or amyotrophic lateral sclerosis (ALS), you may qualify.”
The study will assess the safety of both the wireless chip and the surgical robot used to implant it in the brain. If successful, subjects will be able to control a computer cursor or keyboard using only their thoughts.
The ultimate goal for Neuralink, according to Mr Musk, is to not just treat medical patients but augment the ability of humans in order to allow them to compete with advanced artificial intelligence.
Implanted chips could provide “enhanced abilities” like greater reasoning and improved vision, while enabling people to perform previously impossible tasks like streaming music directly to their brain.
Neuralink has faced criticism from animal rights groups for the treatment of its research subjects at the University of California’s Davis Primate Centre.
The Physicians Committee for Responsible Medicine (PCRM) accused Mr Musk’s startup of subjecting monkeys to “extreme suffering” and providing “inadequate animal care”.
Both Neuralink and Mr Musk have denied the allegations, with the tech billionaire claiming last week that research experiments were only carried out on primates that were already close to death.
“No monkey has died as a result of a Neuralink implant,” he wrote on X. “First our early implants, to minimise risk to healthy monkeys, we chose terminal monkeys (close to death already).”

 Yahoo News
Yahoo News 
