Booster jabs for millions of people to be rolled out from next week
Booster Covid vaccines will be offered to millions of people from next week alongside annual flu jabs.
Those eligible include anyone aged 50 and over, people living and working in elderly care homes, and frontline health and social care workers.
All those who are clinically extremely vulnerable and anyone aged 16 to 65 in an at-risk group for Covid (who were included in priority groups one to nine during the initial vaccine rollout) will also be eligible for a jab.
The Pfizer/BioNTech vaccine will be used as the booster dose for around 30 million people, with experts saying it is safe to be given alongside the usual winter flu jab.
People could be able to get their Covid and flu vaccines on the same day, preferably with one shot in each arm.
Health Secretary Sajid Javid told the Commons that the NHS would contact all those who are eligible and is preparing to offer the jabs from next week.
Children aged 12-15 in England will be offered the #COVID19 vaccination, starting next week.
It follows advice from the UK's Chief Medical Officers. Parent or carer consent will be sought for the vaccination.
Read more: https://t.co/gaPf9Jh8wG
— Department of Health and Social Care (@DHSCgovuk) September 13, 2021
He told MPs: “There’s evidence that the protection offered by Covid-19 vaccines reduces over time, particularly (among) older people who are at greater risk, so booster doses are an important way of keeping the virus under control for the long-term.”
Wales, Scotland and Northern Ireland will join England in offering jabs.
Three vaccines have been approved as safe and effective as boosters – AstraZeneca, Pfizer and Moderna – but experts have decided to opt for Pfizer as a preference after studies showed it is well tolerated and works well as a booster.
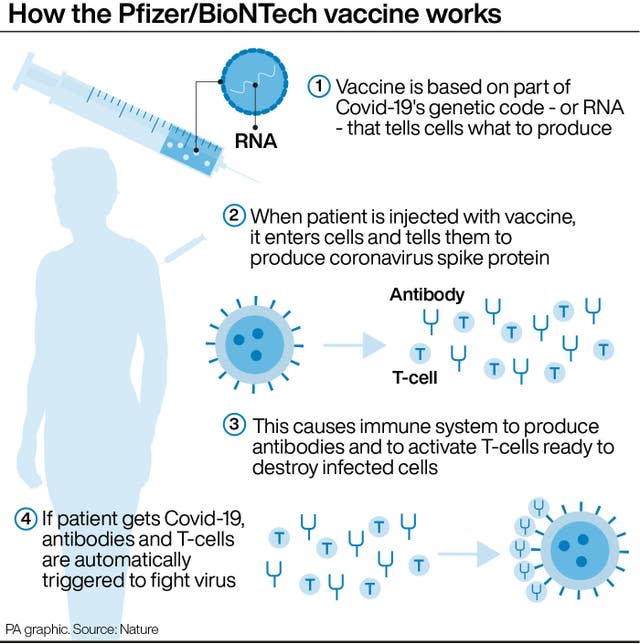
It can be given to people regardless of which vaccine people had previously.
The Moderna jab may be used as an alternative, but as a half-dose booster shot after studies showed it was effective at this dose, with few side-effects.
Moderna has proposed a half-dose for boosters in its applications to European and US regulators.
The Joint Committee on Vaccination and Immunisation (JCVI), who prepared the advice to ministers, said people should receive their third booster dose at least six months after they received their second dose of a Covid vaccine.
When there is more data, the JCVI plans to look at whether boosters should also be offered to healthy people under the age of 50, though there is less concern about immunity waning in this age group.
Professor Wei Shen Lim, chairman of Covid-19 immunisation for the JCVI, said: “The UK’s Covid-19 vaccination programme has been hugely successful in protecting people against hospitalisation and death, and the main aim of the booster programme is to prolong that protection and reduce serious disease as we head towards the colder months.

“The JCVI is advising that a booster dose be offered to the more vulnerable, to maximise individual protection ahead of an unpredictable winter.
“Most of these people will also be eligible for the annual flu vaccine and we strongly advise them to take up this offer as well.”
The advice does not imply there will be a booster programme every six months, he said.
The new guidance from the JCVI appears to differ to its interim guidance published in June.
The interim guidance said anyone over 16 who qualifies for a seasonal flu jab would be included in the booster campaign, which would have included millions of people with asthma.

This has been scrapped, and only those in original priority groups one to nine will be offered a booster, meaning not all those who get the flu jab may be included.
Professor Jonathan Van-Tam, deputy chief medical officer for England, told a Downing Street briefing: “We know that this pandemic is still active, we are not past the pandemic, we are in an active phase still.
“We know this winter could be bumpy at times and we know that winter viruses such as flu and RSV (respiratory syncytial virus) are highly likely to make their returns.
“So with that in mind, the aim of the game – the mantra – is to stay on top of things.”
Prof Van-Tam said that if he was offered the flu jab and a Covid-19 booster at the same time, he would take it.
He stressed that a Covid-19 booster campaign can offer the prospect of a “normal winter life”, adding that a third vaccine will “keep the lid on things Covid-wise”.
When asked why booster jabs are necessary considering there is only a small decrease in immunity months after the second dose, Prof Wei Shen Lim said this small drop in immunity can still lead to a large rise in hospital admissions.
He said: “If we are running at a vaccine effectiveness of, let’s say, 90%, and it drops to 80%, it may not seem like very much.
“But for a certain proportion of people who are being admitted to hospital when vaccine effectiveness is at 90% then you double the drop, you might find that you are doubling the number of people who are admitted to hospital.
“So it does have a big impact because we’re talking about the entire population here and not just a single individual.”
It's really important to discuss the risks and benefits of the vaccine with a healthcare professional."
Dr Edward Morris, president of @RCObsGyn, encourages pregnant women to take expert advice on the #COVID19 vaccination ⏬
More info: https://t.co/wdjafHN1Yx pic.twitter.com/hLxvb6aQ7m
— Department of Health and Social Care (@DHSCgovuk) September 14, 2021
Meanwhile, Prof Van-Tam compared administering booster jabs early rather than waiting for more evidence on post-vaccine immunity to campers who prepare their tents against an incoming storm.
When asked why booster jabs are necessary, considering there is only a small decrease in immunity months after the second dose, he told a Downing Street briefing: “I don’t know if many of you are used to crawling into small tents on mountainsides?
“But if you do so and you know there’s a storm blowing in the night, it’s better to put some extra guy ropes on there and then, than it is to wait until the middle of the night when it’s howling with wind and rain, and you’ve got to get out of your tent and make your tent secure.
“By the time you crawl back in, you’re soaking wet.
“So it’s better to be pre-emptive and to be prepared and plan for the worst possibilities.”
He said the rollout will begin in a “short number of days” through GP surgeries and vaccination centres, adding that the booster programme needed to progress with “good pace”.
Scientists behind the CovBoost trial said that there was a “boost” in antibody levels generated among people who had a third dose in the clinical trial.
The study, which informed the JCVI decision for the booster programme, is due to report its findings publicly in early October.
The trial examined data on 2,833 people at 18 sites across the UK, half of whom had been fully vaccinated with AstraZeneca, half of whom had Pfizer.
Researchers measured side effects and the immune response in the body, including participants’ antibodies and other immunological responses.
Prof Saul Faust, chief investigator of the trial, said: “The data that we’ve been able to collate and provide to JCVI is particularly around the vaccines that we have available for the UK programme.
“We have demonstrated a fold change boost to antibody levels for some of the vaccines, and the cellular responses have been boosted as well.”
He added: “The fold-difference means the amount it has gone up relative to where you started.
“I don’t think it’s fair to talk in more detail because we obviously have seven manufacturers involved in this trial, all of whom provided their vaccines free of charge to and without any intellectual control, and we do as an investigative group have to be responsible, and be fair to everybody in the way we get the data and package it and explain what we’ve seen.
“So with the limited data at the moment I don’t think we can put a number on that for individual vaccines.”

 Yahoo News
Yahoo News 
