First 'variant-proof' Covid vaccine starts trials in Manchester
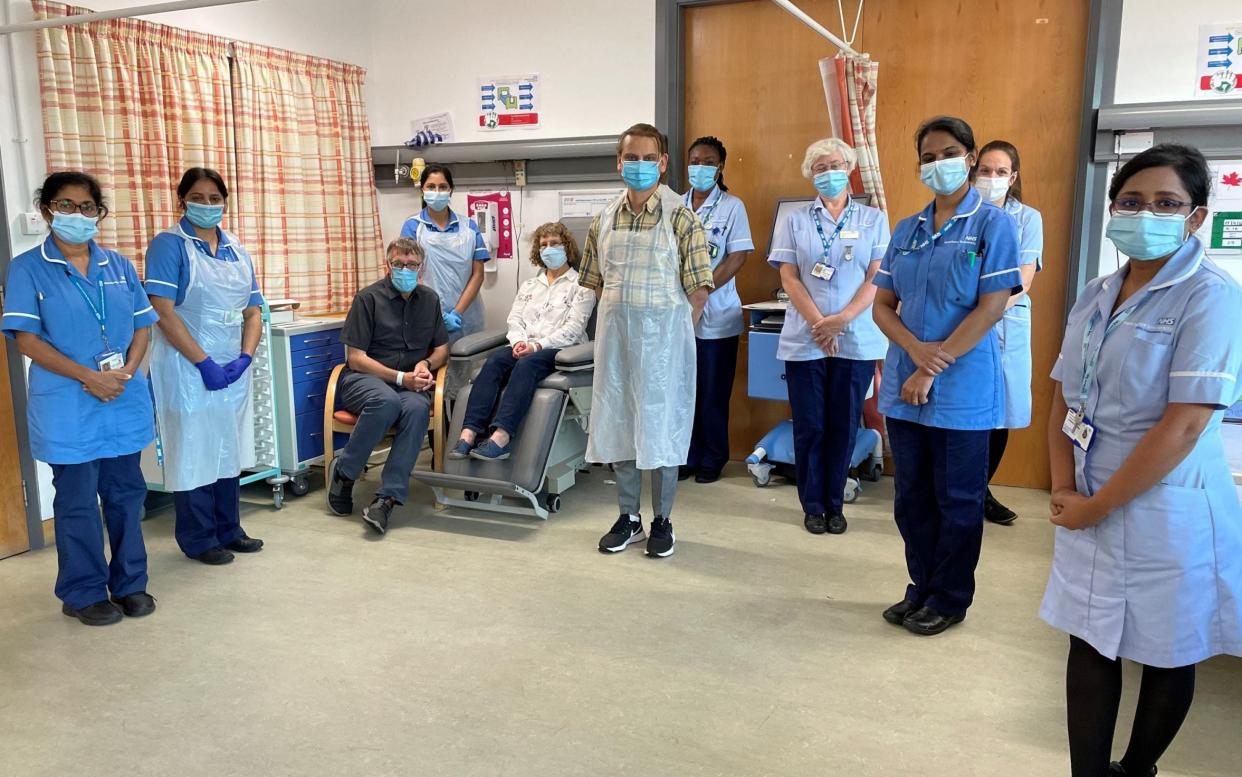
The world’s first variant-proof booster vaccine, which could prevent the need for jabs to be tweaked regularly to combat new strains, is being trialled in Manchester.
Retired couple Andrew Clarke, 63, and his wife Helen, 64, from Bolton, became the first to receive the mRNA vaccine on Monday.
Most current vaccines target the Covid-19 spike protein - the little grappling hook on the outside of the virus that it uses to latch onto human cells.
However many of the worrying new variants have spike protein mutations, which can make vaccines less effective.
As well as spike proteins, the new vaccine - called - currently GRT-R910 - contains other viral proteins that are less likely to evolve over time, and is also designed to induce a strong memory T-cell response.
A phase one trial in the over-60s was launched on Monday by US pharmaceutical company Gritstone in collaboration with The University of Manchester and Manchester University NHS Foundation Trust.
If successful it would be used as a booster to improve the immune response to emerging variants. Results are expected by next Spring.
Professor Andrew Ustianowski, Chief Investigator for the study, from The University of Manchester: "We now know the immune response to first generation vaccines can wane, particularly in older people.
"Coupled with the prevalence of emerging variants, there is a clear need for continued vigilance to keep Covid-19 at bay.
"We think GRT-R910 as a booster vaccination will elicit strong, durable, and broad immune responses, which are likely to be critical in maintaining protection of this vulnerable elderly population who are particularly at risk of hospitalisation and death."
Though the vaccine is being initially trialled in the over-60s, future studies will also examine its efficacy in other-vulnerable populations.
The trial will examine dose, safety, tolerability, and immunogenicity at least four months after the second dose of an initial vaccine.
"Our vaccine is designed to drive robust CD8+ T cell responses, in addition to strong neutralising antibody responses, offering the promise of longer lasting immunity," said Dr Andrew Allen, Co-Founder, President and Chief Executive Officer of Gritstone.
"Since viral surface proteins like the spike protein are evolving and sometimes partially evading vaccine-induced immunity, we designed GRT-R910 to have broad therapeutic potential against a wide array of SARS-CoV-2 variants by also delivering highly conserved viral proteins that may be less prone to genetic variation in the virus.
"Our hypothesis is that a different vaccine such as GRT-R910 might complement the primary immune response from pre-existing vaccination with a first generation Covid vaccine in such a way that it would provide more benefit than an additional dose of the same vaccine."
The Clarkes, who are both fully dosed, said they wanted to 'play their part' in ending the pandemic.
Mr Clarke added: "Somebody has to be the first and we’re confident in the science and technology behind this vaccine and convinced of the need for it.
"Because we’re both retired, we feel we had a reasonably easy lockdown, but we know it wasn’t the same for everybody.
"We feel that this is perhaps a small part we can play in helping to make things change."

 Yahoo News
Yahoo News 
