Mixing Covid vaccines 'may give better long-term protection'

Mixing vaccines could improve protection against coronavirus, a senior government scientific adviser has said.
Prof Anthony Harnden, deputy chairman of the Joint Committee on Vaccination and Immunisation, said different vaccine types could coalesce to boost the immune system and provide a longer-lasting response. He also said a "mix and match" approach could make future rollouts more flexible.
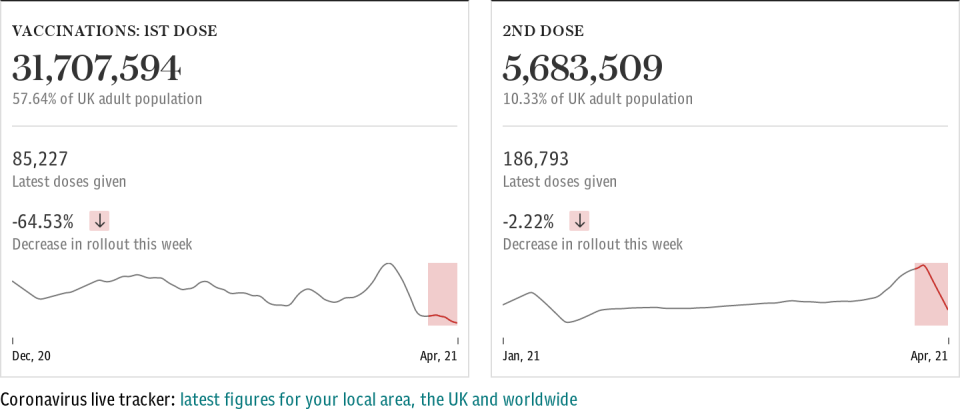
It comes after planners were forced to rethink phase 2 of the rollout, which started on Tuesday, after the JCVI recommended people aged 18 to 29 have an alternative to the AstraZeneca jab due to blood clotting concerns. Currently only two other vaccines - Pfizer and Moderna - have received regulatory approval in the UK.
The promise of 30 million one-jab Johnson & Johnson doses in the summer suffered a setback this week after US regulators suspended its use, also due to fears over clotting.
Prof Harnden said: "If we can mix and match, we'll get much better flexibility - we will be able to potentially use different vaccine booster campaigns in the autumn. And, in fact, mixed schedules may - and this is a big may - but they may give better longer-term protection, and that will be very interesting to see."
His comments followed the announcement of a new trial to test the effect of mixing vaccines.
Led by Oxford University, the Com-Cov2 study will assess the immune response of people who have had two of the Pfizer, AstraZeneca, Moderna and Novovax jabs.
While the Government has said that people should get two jabs of the same vaccine in the current rollout, a finding that mixing is beneficial, or at least not inferior, could greatly improve the flexibility of a third "booster" jab rollout, which could take place in the autumn.
Asked about the pausing of the Johnson & Johnson vaccine in the US, Prof Harnden said that regulators examined the safety data "almost on a daily basis".
He told Good Morning Britain: "It is of concern, and we will keep a very close review of the situation."
Watch: Mix and match vaccine study extended

 Yahoo News
Yahoo News 
