Normal blood testing to be resumed after NHS supplies bolstered by US stocks
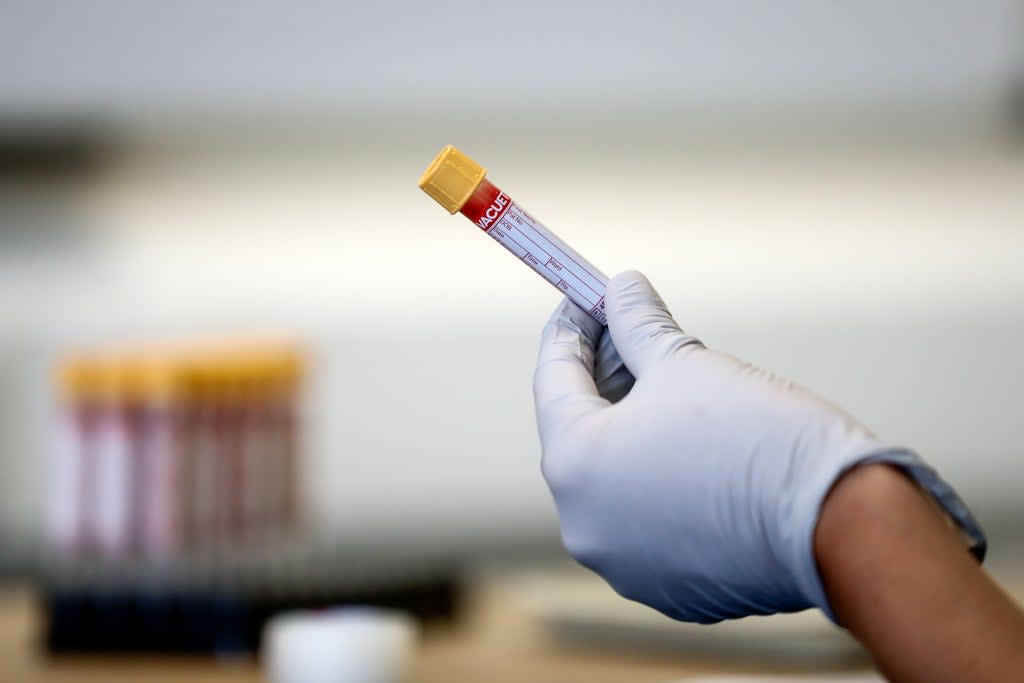
Non-urgent patients in England and Wales should be able to get blood tests when required following reinforcements of much-needed tube supplies, NHS England has said.
A shortage of the tubes from US supplier Becton Dickinson (BD) had forced the NHS in these nations to suspend all non-urgent blood testing last month.
From August 26, only high-priority patients, including those with suspected sepsis or conditions with a risk of death or disability, or those which were “extremely overdue” have been able to get blood tests.
This meant hospitals had to cut blood testing by one quarter.
But a trio of NHS England directors announced in a letter to GPs and trusts that imports of nine million tubes from BD’s US stocks mean that normal testing is likely to be resumed from October 8.
These developments, alongside the efforts of NHS staff to manage use, mean that the supply situation is no longer as constrained
Letter from NHS England directors
The letter from NHS England national medical director Professor Stephen Powis interim chief operating officer Mark Cubbon and medical director for primary care Dr Nikita Kanani warned that the supply issue is “not yet completely resolved”.
The letter said: “Becton Dickinson has now completed improvements to its manufacturing facility in the UK and expects its production capacity to recover through September.
“These developments, alongside the efforts of NHS staff to manage use, mean that the supply situation is no longer as constrained… however, the issue is not yet completely resolved.”
NHS England added that primary and community care services, which include GP surgeries, can resume normal testing from Friday, and that A&E departments, community hospitals and mental health trusts must maintain lower levels of testing until October 8.
The tubes from US stocks have been approved for use in the UK by the Medicines and Healthcare products Regulatory Agency (MHRA).
Read More
Ambulance crisis caused by more than pandemic, senior surgeon warns
PPE worth £2.8bn is not fit for purpose, health minister admits
Scientists trialling whether children need second coronavirus vaccine

 Yahoo News
Yahoo News 
