How the UK's vaccine gamble paid off – and the EU left itself without a leg to stand on

Back in March, the University of Oxford was on the brink of signing a deal with the German drugs giant Merck to research and develop a coronavirus vaccine.
The Government stepped in and helped steer Oxford towards a partnership with British-based AstraZeneca instead, taking a huge gamble by helping to fund the research and development of a vaccine that might have proved useless.
Instead, the Oxford vaccine became only the second in the Western world to be approved for use, and both the UK and AstraZeneca are now reaping the benefits of the deal that was struck last April.
AstraZeneca has, so far, remained fiercely loyal to the Government, resolutely refusing to give in to EU demands that it should redirect supplies of its UK-made vaccine to the bloc.
But that close relationship has come under unprecedented strain as the company found itself under huge pressure from Brussels.

The row erupted last Friday, when AstraZeneca said it would not be able to fulfil promised supplies of the vaccine, which is expected to be authorised by the European Medicines Authority at the end of this week.
Brussels had ordered up to 400 million doses in total and paid €336 million (£296.4 million) in advance to secure its supply of the AstraZeneca vaccine, but the company said it was facing production problems in its EU plants.
EU officials said AstraZeneca had only offered a quarter of the 100 million doses it had promised for the first quarter of the year, while the firm put the figure at 31 million out of 80 million jabs it had hoped to deliver.
The EU's 27 member states, many of whom had foregone other vaccines to wait for the AstraZeneca jab – which is cheaper and easier to store than the Belgian-made Pfizer alternative – were furious and wanted answers.
The European Commission, which had negotiated its own contract with AstraZeneca, was left red-faced and angry. And so the blame game began.
On Monday, the commission subjected AstraZeneca to a string of public and private dressing downs, accusing it of breaching its contractual obligations.
The commission ratcheted up the pressure, saying it would force drugmakers in the EU to ask permission to export vaccines out of the bloc. The threat to British supplies of the Pfizer vaccine was clear.
On Wednesday, the EU went further, demanding that AstraZeneca divert British-made supplies of the vaccine to the bloc.
Watch: COVID-19 - EU eyes UK vaccine supply as AstraZeneca row deepens
The British Government has a deal with AstraZeneca to supply 100 million doses of the AstraZeneca vaccine. Crucially it also said Britain's two plants could only supply jabs outside of the UK once that order was fulfilled.
That agreement was struck three months before the EU's deal as ministers took advantage of their post-Brexit freedom to go it alone rather than signing up to EU-wide procurement.
While Britain was setting up its logistics and sorting out supply chains, the EU's negotiations with AstraZeneca became mired in bureaucracy, costing the bloc vital time which has now been manifested in its supply problems.
On Tuesday, the AstraZeneca CEO, Pascal Soriot, said separate supply chains, including manufacturing plants, had been created for each order – one for the EU and one for the UK. This was done explicitly to stop squabbling over supply.
The EU supply chain was behind because the bloc, late to the party, was still experiencing teething problems with production – problems the UK supply chain had also suffered initially but had overcome.
Mr Soriot added that AstraZeneca was under no contractual obligation to supply the vaccines but only to make its "best efforts" to meet the order. His interview – in which he pointed out that the vaccines were being supplied on a not for profit basis – further infuriated the European Commission top brass.
Brussels has made huge play of the virtue of negotiating as a bloc, a posture that has appeared increasingly ridiculous as it lagged behind Britain in the vaccination rollout.
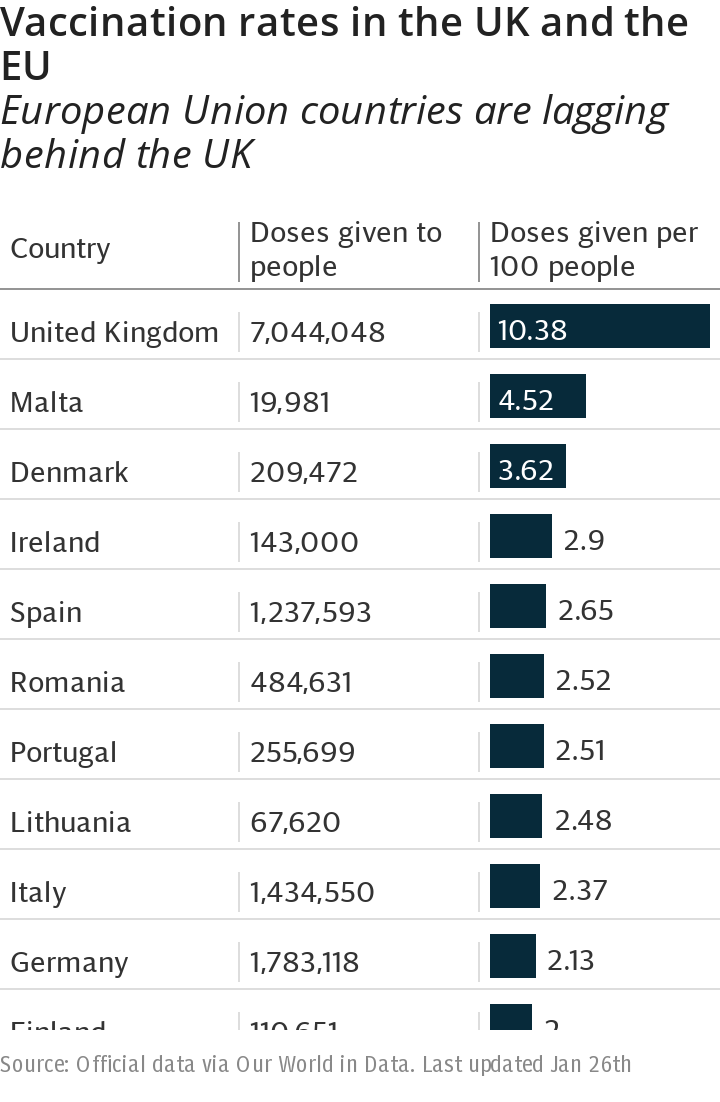
To make matters worse, the EU member states have been suffering from logistics and delivery problems for the other two vaccines already approved.
The Government was invited to join the EU joint procurement system but declined and made use of emergency authorisation procedures, open to all EU member states, to speed up approval of vaccines. Brussels sneered that it preferred not to take such risks.
Mr Soriot's implication that the EU's dithering was to blame for the delay came on the eve of EU countries such as Spain running out of vaccines and having to pause their programmes.
On Wednesday, the commission went back on the attack. It accused Mr Soriot of revealing confidential information, such as the "best efforts" clause and the production capacity, in his interview. Legal action was threatened before the threat was later played down.
Stella Kyriakides, the EU's health commissioner, told reporters in Brussels: "We reject the logic of first come, first served. That might work at the neighbourhood butchers, but not on our contracts and not in our advanced purchase agreements."
Ms Kyriakides said there was "no priority clause" in the EU contract between the four production plants in the agreement, which include two factories in the EU and the two in the UK, adding: "In our contract, it is not specified that any country or the UK has priority [...] this needs to be absolutely clear."
The European Commission's chief spokesperson said: "If UK plants are working better, are we expecting UK plants to deliver doses to the EU? Yes we do. "
Elsewhere in Brussels, AstraZeneca's name continued to be dragged through the mud. There was talk of a nascent vaccine trade war from one German MEP.
Watch: AstraZeneca facility closed after bomb scare
Ms Kyriakides turned the knife and played to the gallery; making the reputational risk facing AstraZeneca in the EU crystal clear. She said the EU was losing people to the pandemic "every day" and added: "Pharmaceutical companies, vaccine developers, have moral societal and contractual responsibilities which they need to uphold."
In the heightened political drama, it mattered little that AstraZeneca is already right on the limit of what is possible. The 100 million doses destined for Britain is what would normally be produced in a year rather than each month.
AstraZeneca stayed firm and loyal to the UK, justifying the Government's decision to move first and fast. But the company remains under huge, sustained legal, financial and reputational pressure from the EU, which is a much larger and more lucrative drugs market than Britain.
The fear is that eventually it will yield to EU demands and redirect supply, now or in the future, but the firm insists it has no intention of doing so.
After another meeting with EU leaders on Wednesday night, a spokesman for AstraZeneca said: "Our CEO was pleased to participate in a meeting with the EU's vaccine steering board this evening. We had a constructive and open conversation about the complexities of scaling up production of our vaccine, and the challenges we have encountered.
"We have committed to even closer co-ordination to jointly chart a path for the delivery of our vaccine over the coming months as we continue our efforts to bring this vaccine to millions of Europeans at no profit during the pandemic."
In an ironic post-script, earlier this week Merck – the firm that had come so close to signing up with the University of Oxford – announced that it was abandoning its attempts to develop a vaccine after disappointing results in trials of two prototypes.

 Yahoo News
Yahoo News 
